Utilities
The utilities system inside an ATMP manufacturing facility—particularly its HVAC design—plays a very significant role in managing the risk of cross-contamination.
Again, working on the frontier of precision medicine means there’s no template to follow. Manufacturers and their project teams must develop a tailored approach to integrating the mechanical, electrical and plumbing (MEP) elements necessary to balance biosafety with GMP expectations in an environment that relies on highly specialized—and infectious—materials. Every detail of the utilities system comes into play, but three elements are especially critical:
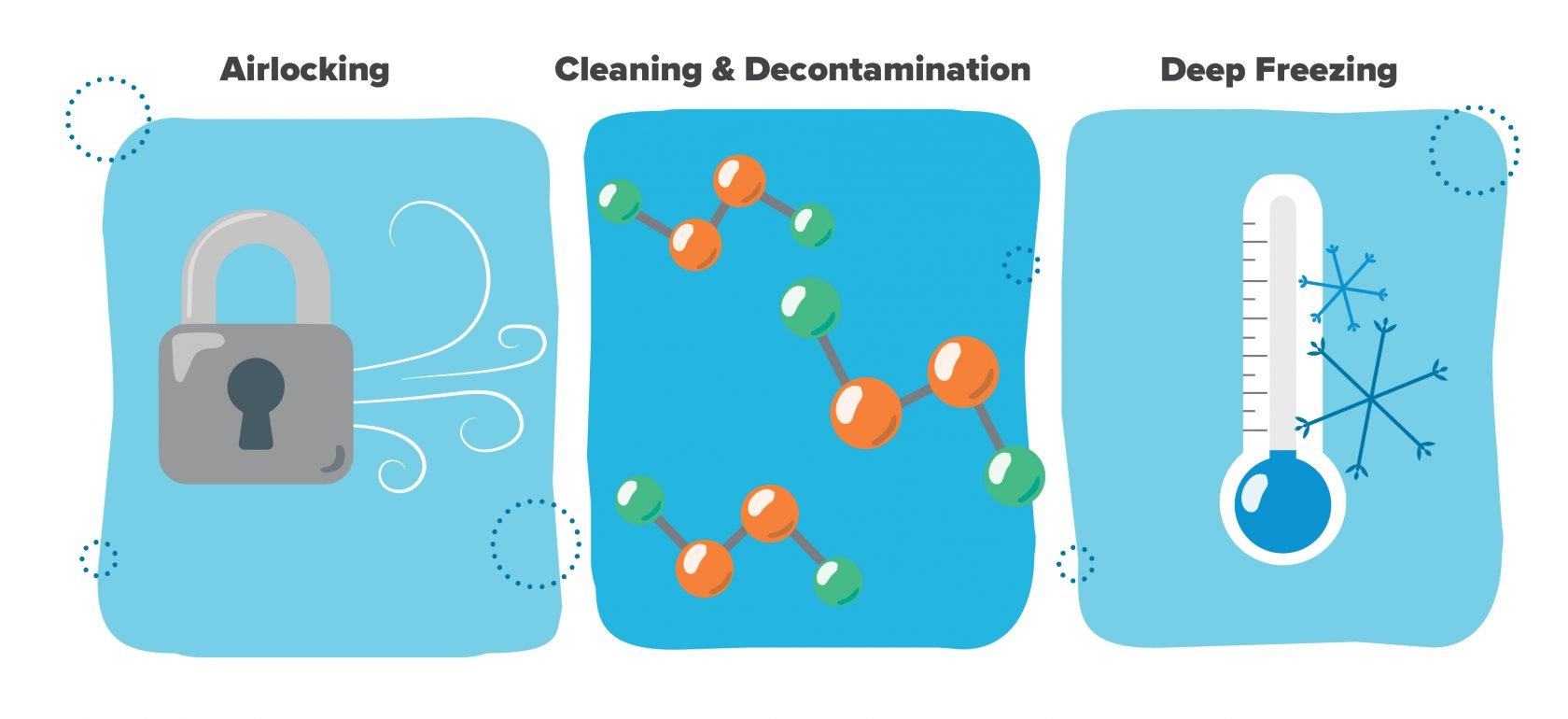
Airlocking
To ensure the safety and future flexibility of the ATMP manufacturing environment, manufacturers need an airlocking strategy designed to facilitate the flow of personnel and materials while reducing the potential for cross-contamination.
One of the best ways to approach this challenge, particularly when designing a facility outside of the well-understood parameters of traditional biopharma production, is through computational fluid dynamics (CFD). Using a digital twin of the real ATMP manufacturing environment, project teams can leverage CFD to run scenarios that identify contamination risks and their solutions, which is invaluable when tailoring a strategy for optimal containment, segregation and product protection.
Cleaning and decontamination
This comes down to a facility’s particular risk management approach. Will you require a proactive, templated protocol for managing risk, such full decontamination via vaporized hydrogen peroxide (VHP)—or can you adequately manage risk with a simpler, more reactionary approach, such as a chemical wipe-down in the event of a spill?
Often, choosing an appropriate cleaning strategy is a scenario-based decision. VHP decontamination is costly and takes time, but ATMP manufacturers—particularly those working with viruses—are typically more inclined towards this cautionary and highly effective decontamination approach.
The facility’s risk management approach should also consider the flow of cleaning personnel and their equipment, ensuring that they do not become vehicles for cross-contamination. This could mean designing segregated pathways for cleaning staff, and/or storing cleaning materials in product-differentiated areas outside of the production core.
Deep freezing
Depending on the nature of their products, many ATMP manufacturers require high volumes of liquid nitrogen to supply a network of controlled rate freezers and cryo freezer storage.
Storing and distributing liquid nitrogen involves complex site logistics and careful design. Vacuum-insulated piping is expensive, so the distance between your bulk liquid nitrogen tanks and your freezers will quite literally cost you. This, too, should impact your approach to facility design.
In some cases, such as when designing facilities with limited space, ATMP manufacturers might rely on portable cylinders instead of bulk tanks, but this too can set off a complex chain of design issues. First, there’s the question of setting up a dedicated shipping and receiving area for the cylinders, along with a robust tracking system. Then there’s the question of getting all of those cylinders where they need to go, safely and efficiently. Liquid nitrogen can asphyxiate a human in close quarters; that rules out the possibility of relying on elevators, or else it requires a way to move cylinders on and off of elevators with no one there to assist.
ATMP manufacturers who rely on deep freezing to preserve the stability of their products need a well-considered facility design that accommodates the safe storage and distribution of liquid nitrogen.
What’s next in ATMP manufacturing?
While navigating the maze of unknowns that stretches before them, ATMP innovators can be sure of one thing: solutions will emerge. The cost and complexity of manufacturing cell and gene therapies will taper as technology catches up and emerging performance data teaches us how to optimize and improve tomorrow’s facilities.
We can say all of this with certainty, because we’ve seen it before: in the 1970s and 1980s, the early biotech innovators were at work, forging a new path without a map to follow. Just like today’s ATMPs, the monoclonal antibody therapies (mAbs) developed thirty or forty years ago relied on novel technologies and were, at the time, prohibitively expensive and the target of much skepticism. Today, mAbs are a standard feature of drug manufacturing—it’s hard to imagine modern medicine without them.
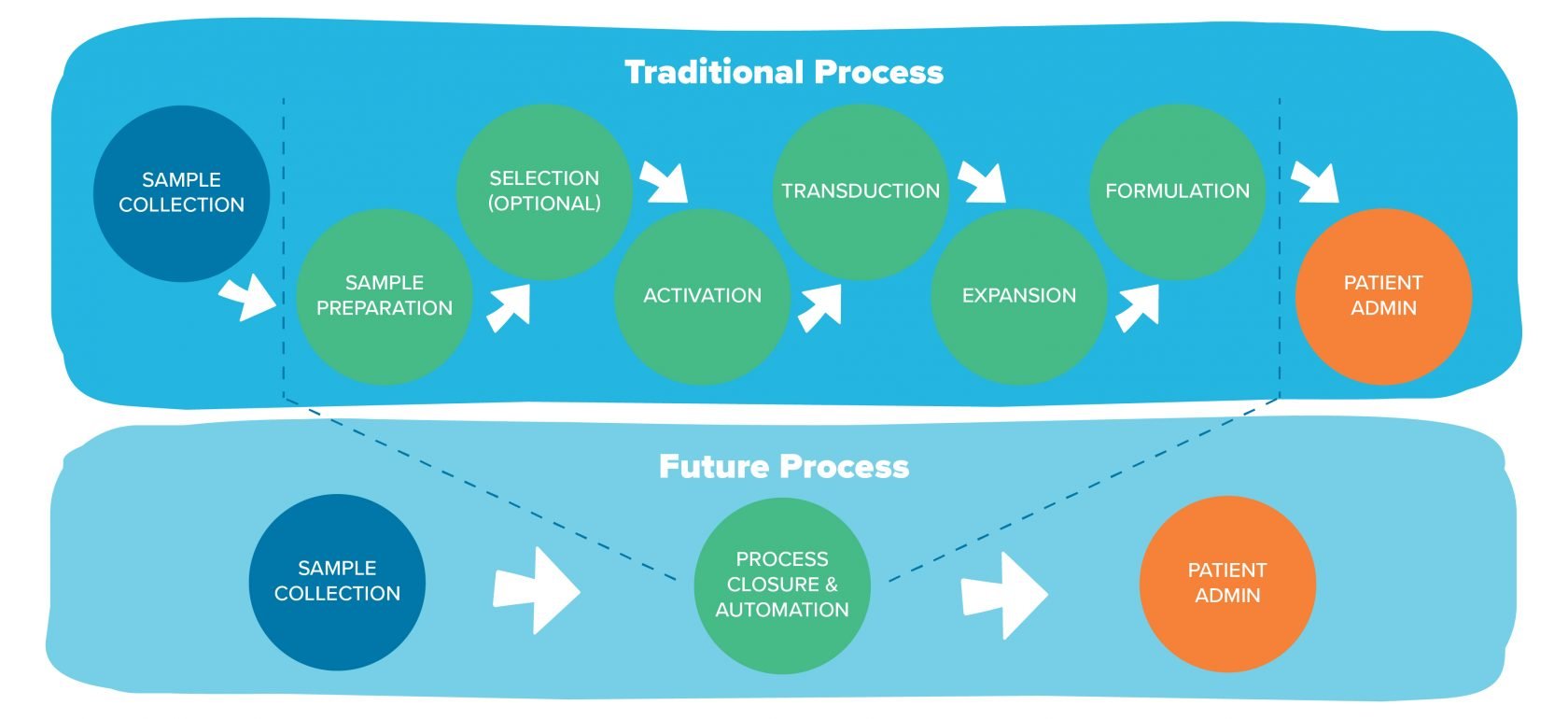
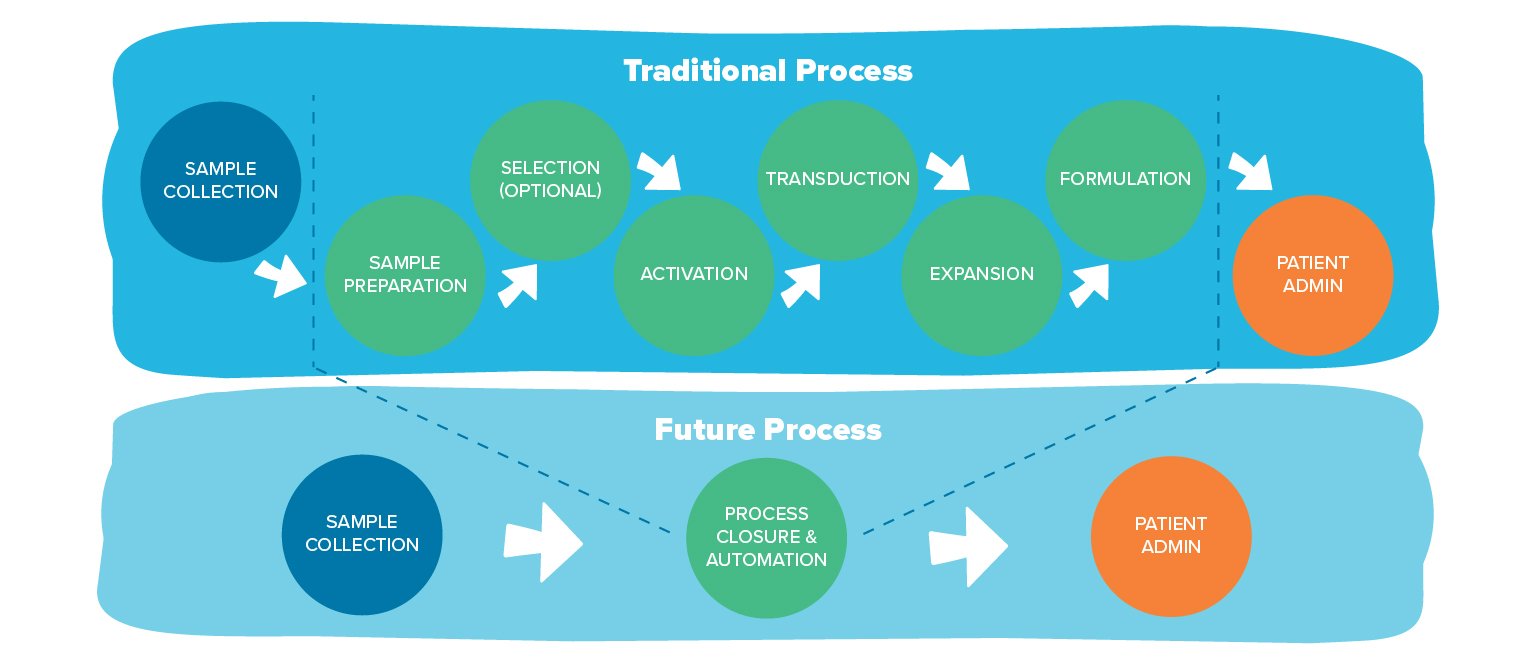
The same story will play out in ATMP manufacturing. Many factors will converge to get us where we need to go, but the emergence of three particular innovations will play a key role: