An in-depth review of what a monoclonal antibody is, how it’s produced and the challenges the industry faces.
Read More
Along with reductions in equipment size and scale, and reduced facility footprint, continuous chromatography helps companies drive down raw material costs, sustain a mindset of continuous improvement, and meet their sustainability goals.
Why is process intensification important for biomanufacturers?
As the number of approved biopharmaceutical and synthetic biomolecules continues to rise to meet growing patient demand, companies will be under increasing pressure to reduce cost of goods through process intensification. As legacy products, particularly monoclonal antibodies (mAbs), see increasing competition from biosimilars, reducing the cost of goods becomes paramount.
High-titer cultures put pressure on downstream processing
Process intensification has gained prominence, in part because the titers of mammalian cell cultures (e.g., CHO cells) have increased dramatically over the past three decades. From an upstream process perspective, increasing cell culture productivity means facility throughput targets can be achieved using smaller bioreactors or even single-use equipment, and thus a more compact facility footprint.
Increases in upstream productivity require scale-up of downstream unit operations to support commercial manufacturing. Batch chromatography columns must be scaled up to house enough resin to purify the additional material and quickly become heavy and difficult to transport. And, once columns are at the maximum size, increasing purification capacity is only possible by adding chromatography cycles, which can create a scheduling bottleneck. For these reasons, batch chromatography can become rate-limiting to the overall facility run rate.
An intensified chromatography process is predicated on continuously operating a set of smaller columns and a smaller fluid handling unit. Continuous chromatography is already in use for peptide and oligonucleotide purification. It’s unlikely, however, that facilities already built for large-scale batch chromatography will be retrofitted with continuous chromatography equipment.
Terms to know
A philosophy or approach aimed at increasing the specific productivity of a manufacturing process. An intensified process enhances productivity by increasing the ratio of product mass-produced per unit mass of consumed raw material. One way this can be accomplished is by increasing product titer in a cell culture bioreactor. Another is through the application of continuous chromatography methods.
Cell culture titer is defined as product concentration in the bioreactor – specifically, the mass of product expressed per liter of cell culture volume.
A bacterial recombinant protein possessing high affinity for the fragment crystallizable region (Fc region) of monoclonal antibodies and fusion proteins. Protein A immunoaffinity chromatography is ubiquitous in the biopharmaceutical industry as the primary step in mAb purification processes.
The product recovery step of a “bind-elute” chromatography process. During the elution stage, the product of interest is separated from other species via differential migration through the packed bed (stationary phase). Components bound to the stationary phase are eluted in order of least-retained to highest-retained. The concentration of the eluting modifier may be either constant (i.e., “isocratic”, or “step gradient”) or varying (i.e., “gradient”) during the elution process.
Product-containing solution collected as column effluent during the elution stage of the chromatography process.
A continuous liquid chromatography platform in which the mobile phase composition is isocratic (constant composition). Intermittent counter-current motion of solid (stationary phase) and fluid (mobile-phase) phases result in continuous separation of a binary mixture. In contrast to TMB (True Moving Bed), where solid and fluid phases theoretically move in continual counter-current motion, SMB operates in a periodic steady state. Simulation of the “moving” bed occurs by switching the inlet and outlet streams connected to each column at a specified frequency. Solutes contained in the SMB feed stream exit the system in the extract and raffinate streams.
An isocratic separation method in which differential migration of solutes in the mobile phase occurs based on molecular size. The stationary phase particles contain a specified range of pore sizes but contain no functional group derivatization. The result is that there are no effects on chromatographic peak shapes or peak resolution arising from adsorption equilibrium. In SEC applications, the rates of solute migration are based on molecular size. Small molecules permeate the adsorbent pore structure to a much higher degree than larger molecules and therefore migrate more slowly. Size exclusion chromatography may also be used to facilitate buffer exchange, since macromolecules move ahead of the buffer front. Thus, the product of interest is always eluted in equilibration buffer, regardless of the feed background buffer composition.
A continuous liquid chromatography platform in which implementation of two or more columns facilitates uninterrupted feed flow. In a multi-column MCC platform, resin contact efficiency is increased by allowing feed “breakthrough” onto a second column in the loading zone. In principle, a minimum of two columns are required for MCC. However, the more efficient MCC system typically consists of three or four columns.
A continuous liquid chromatography platform in which yield and purity of a ternary separation both increase due to internal “side-cut” recycling of impure pre- and post-peak fractions. Note that despite inclusion of the term “counter-current,” all continuous liquid chromatography methods simulate counter-current action between the mobile and stationary phases. The term “solvent gradient” alludes to the separation difficulty associated with purification of full-length synthetic molecules, such as peptides and oligonucleotides.
Real-time or near-time analytical monitoring designed to enhance control and reproducibility of biomanufacturing processes. The United States Food & Drug Administration first published guidance on the PAT initiative in 2004, as part of a new emphasis on Quality by Design and Quality Risk Management.
An in-line technique used to measure how a beam of incident light by a continuous sample stream. The range of scattered light intensities may be used to determine the average size of molecules contained in the stream.
What is batch chromatography?
Conventional batch liquid chromatography operations are based on sequential, repeated cycles of purification. A portion of feed containing the product of interest is purified during each cycle, with product eluate collected into a single pool vessel. A single chromatography column is used, which must be sized to ensure that the complete set of cycles is completed in time to be ready for the next batch of feed material. As such, typical batch operation requires large chromatography columns, high pumping capacity, and storage vessels large enough to accommodate the complete volume of purified product.
For the overall downstream purification of a mAb, a batch process requires the completion of one unit operation before the next begins. Typically, three liquid chromatography steps are required to achieve the product purity required for the final formulation of bulk drug substance. Commercial-scale biomanufacturing has titers and run rates that typically drive the use of large-diameter columns that result in larger chromatography skids, utility infrastructure and controlled spaces.
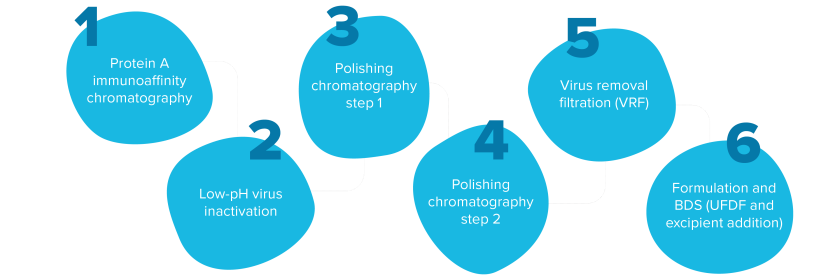
The typical sequence of unit operations in a downstream mAb purification
What is continuous chromatography?
Unlike batch chromatography, continuous chromatography proceeds without interruption and is ideal for large-scale production due to its efficiency. Continuous liquid chromatography can be paired with fed-batch or continuous perfusion cell culture. Product eluate is collected continuously or semi-continuously, and adjacent unit operations are allowed to overlap. Continuous capture chromatography can be integrated with continuous upstream perfusion cell culture to match the output of a conventional batch manufacturing process—with considerably reduced equipment outlay.
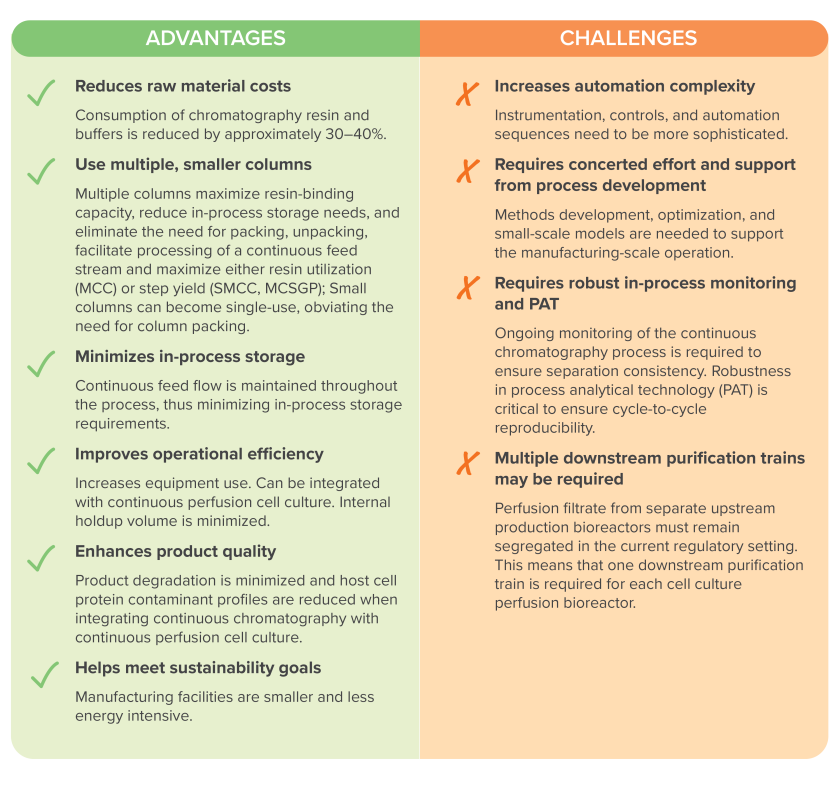
Continuous chromatography platforms
Different types of continuous chromatography platforms can be applied in downstream biomanufacturing, including:
Simulated moving bed (SMB) chromatography
SMB chromatography is the oldest continuous chromatography platform but is relatively unknown in the biopharmaceutical industry. Of all the continuous chromatography platforms, it’s the only one that features true steady-state operation—feed and product eluate streams flow into and out of the system at constant flow rates. Size exclusion chromatography (SEC) is typically only used as a polishing chromatography step as a last resort, due to limitations in throughput rate and logistical issues with resin handling. However, SEC represents one potential application of SMB chromatography.
Multi-column capture (MCC) chromatography
MCC chromatography increases resin use by loading the column beyond the point of product breakthrough. A second column, connected in series with the overloaded column, captures product in the breakthrough effluent, thus maximizing product recovery and equaling the yield of the conventional batch process. Three to eight columns can be used in an MCC application, depending on the process recipe and the relative durations of the loading and recovery stages. We detail a three-column MCC configuration in the case study developed in the downloadable white paper. A four-column MCC configuration represents the most common scenario, which is represented in the below animation.
Continuous chromatography with side-cut recycling
In both conventional batch chromatography and MCC chromatography, product is lost in the pre-peak and post-peak fractions (i.e., side-cut fractions) during collection of the main product peak (i.e., center-cut fraction). Two separate applications of continuous chromatography are designed to increase chromatographic step yields via internal recycling of the side-cut fractions. Sequential multi-column countercurrent (SMCC) chromatography and multi-column solvent gradient purification (MCSGP) are more complicated than MCC chromatography and require significant time for method development and optimization.
The animation depicts a four-column multi-column capture chromatography process. The sequence is based on the example depicted in the white paper, where the repeating cycle duration is 7.2 hours. The animation runs for two minutes, depicting two complete cycles (a total of 14 hours and 40 minutes of “real-time”). During each cycle, each of the four columns proceeds through a complete batch recipe. The elements of the batch recipe are pictured in the schedule excerpt shown below.
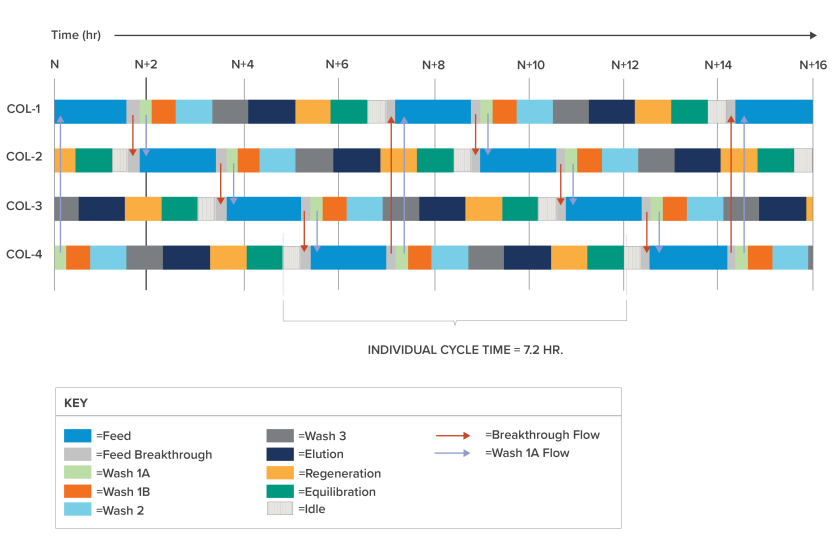
Note first that feed flow is continuously supplied to the system, with the feed input switching to the second column once product “breakthrough” occurs from the upstream column. Once a column has completed its feed stage it is subjected to a series of wash steps, preparing the column for the elution stage. Note that a concentrative effect occurs as the product fraction (green) is separated from other bound impurities and collected into the dedicated product pool vessel.
Following elution, the column is regenerated to remove residual contaminants and then re-equilibrated to prepare for the next feed stage. At this point the batch recipe associated with the selected column is complete. One cycle of the four-column MCC process is complete when the batch recipe has been executed once for each of the four columns.
Continuous chromatography in action
A three-column multi-column capture chromatography operation integrated with continuous perfusion cell culture
There’s a gap that needs to be bridged to successfully reap the benefits of using continuous chromatography as part of your process intensification strategy. While the fundamental operating principles of continuous chromatography platforms may be easy to understand, development and implementation requires expertise and concerted effort. All parties involved in biopharmaceutical production can expect new challenges associated with the advent of continuous operations and process intensification.
Our white paper includes a case study for process intensification, in which a three-column MCC chromatography platform is integrated with a continuous perfusion cell culture process. The analysis reveals that two 5,000 L perfusion bioreactor trains, each accompanied by a dedicated MCC chromatography system, are capable of equaling the productivity of six 10,000 L fed-batch bioreactors operating at a facility run rate of 3.0 batches per week.
The intensified process resulted in a 30% reduction in the usage of costly protein-A immunoaffinity resin and buffer components, leading to annual savings of $4 million specifically in protein-A resin expenses during production. For a detailed examination of this case study, click here.
While the case study details a viable scenario leveraging continuous capture chromatography that would result in considerable cost savings for mAb production, even greater process intensification is possible. We outline forward-looking scenarios that could reduce cost of goods by another order of magnitude, by targeting the most expensive raw material in mAb downstream processes—protein-A immunoaffinity resin—for replacement with conventional resins.
Continuous chromatography will shape the future of biomanufacturing
Process intensification is complex and requires considerable resources to achieve. Improvements in real-time PAT, including spectroscopic techniques like near-infrared spectroscopy and real-time multi-angle light scattering (RT-MALS), will add to the cost and complexity. While the development, engineering, and validation of continuous chromatography operations can be daunting, manufacturers must consider the cost of not pursuing process intensification.
The future of biomanufacturing appears to be rooted in continuous processing, which is coupled with an opportunity to reduce raw material costs, improve process efficiency, and achieve higher product quality. In addition, a substantial reduction in equipment size and capacity simplifies facility design and construction and aligns with many corporate sustainability goals.
Want to learn more about continuous chromatography?
Download our continuous chromatography white paper to get started or talk to our team of experienced subject matter experts to take your first step toward an intensified chromatography process.

Which is more sustainable: stainless steel or single-use systems?An in-depth analysis of the environmental impacts of single-use technology and stainless steel systems in pharma and biotech manufacturing.
Read More
An in-depth analysis of the environmental impacts of single-use technology and stainless steel systems in pharma and biotech manufacturing.
Read More