
Spray drying is a rapid, robust, reproducible, and scalable process.
First invented in 1872, it became widespread during WWII as the dairy industry embraced the process to produce powdered milk for the military. Shortly after, spray drying was introduced into drug substance and drug product manufacturing. Over time, it has been applied in numerous applications for poorly soluble compounds and high drug-loaded products.
Spray drying is a pharmaceutical process to create dry powder out of a liquid solution, and is used in the production of numerous dry powder products including active pharmaceutical ingredient (APIs) penicillin, enzymes, excipients, bulk agents, controlled drug release formulations, and more. It offers consistent particle size, long-term stability, and increased bioavailability—to mention just a few benefits. Thermolabile—easily destroyed or changed by heat—APIs and dry powder formulations/aerosols also benefit from this technique, since it allows them to be dried at atmospheric pressure and low temperature. But given the nature of the process, it requires unique facility considerations to plan for multi-level equipment, as well as material storage, handling, and containment to protect product and personnel.
Advantages of spray drying
There are many reasons why a pharmaceutical facility might consider spray drying operations:
- Automation-friendly
- Effective control of particle size, shape, form, moisture content, and other properties
- Large output capacities with a wide range of production rates
- Semi-continual production
- Design flexibility
Parts of a spray dryer
While exact design details of a spray dryer vary by specific application and vendor, all spray dryer models have the same basic elements:
- Filtered air inlet
- Air heating source
- Liquid feed and pumping with atomizer
- A drying chamber
- Primary collector with powder recovery/separation
- Outlet air or recirculation system
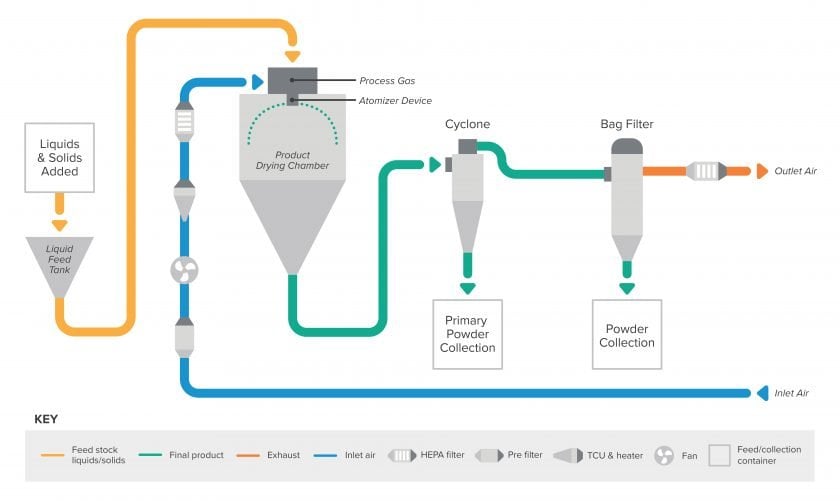
Spray drying process
Spray drying is a semi-continuous process of spraying a liquid solution (with dispersed powder) into a hot air chamber to evaporate the volatile fraction and produce a very fine powder. Most spray drying manufacturing processes are primarily driven by gravity feeding of equipment, thus requiring a multi-level, through-the-floor tower configuration. When designing or renovating a spray drying facility, there are important considerations throughout the entire process.
Receiving materials
Once incoming materials arrive at the receiving dock, they are typically sampled, quarantined until a sample is approved, and stored in appropriate warehouse conditions. Incoming materials may come in the form of various dry powders and liquids (aqueous and/or solvent). For large volume applications, a facility may employ a liquid tank farm.
Weigh/dispense
After incoming raw materials are released from quality control, dry powder ingredients may be kitted in a weigh/dispense area into appropriate process feed containers (e.g. IBCs, drums, SuperSacks, etc.). The weigh/dispense process is typically done in a downflow booth that provides scales and other apparatus for accurate weighments, as well as dust control. For any highly potent or hazardous materials, additional containment controls and technologies are often applied.
Space planning for weigh/dispense
When planning out the space required for weigh/dispense, it’s important to consider the following:
- Good adjacency and minimum travel distance from warehouse
- Adequate incoming material staging space for good control and elimination of any potential material mix ups
- Controlled weighing/container charging via downflow booths or containment isolators with appropriate material handling for safe ergonomic handling
- Adequate final weighed kit material staging space for good control and elimination of any potential material mix ups
- Appropriately sized work areas
Solution tank feed
After weigh/dispense, dry powders are loaded into the spray drier liquid feed tank(s), mixed appropriately to create a workable suspension, and pumped into the top of the spray dryer where the liquid is atomized and the spray drying process occurs.
The spray dry process allows the liquids to be converted into very fine powders via an evaporation process which are then collected as the final product. This may occur via collection in a cyclone separator and/or a filtered bag house. The final product characteristics depend on the design/operation of the spray dryer and the physicochemical properties of the feed.
Dryer packout and feed
After the spray dried powders are collected, they may undergo additional processing in the form of any post-drying processes, sifting/sizing, and/or additional packout into final containers. The final product is ultimately returned to the warehouse, which may include any cold storage or other environmental conditions, as required.
Spray drying facility considerations
Spray drying facilities can set themselves up for long-term safety and success through thoughtful process and facility planning. As part of your planning considerations, review the implications of product, personnel, and waste flow optimization. Also give consideration to the fact that spray drying is a multi-level process, and has unique needs when it comes to collection container choice, cleaning, hazard containment, and contamination prevention.
Multi-level facilities
When designing a multi-level facility, it’s important to consider the cGMP access between all operating levels. For example, if the spray dryer requires multiple stories for the vertical stack of equipment, then every level should offer operator access—with or without personnel and material airlocks per the facility design and space classifications. In addition, internal GMP staircases can provide access to the entire process space, allowing operators to traverse all levels without having to disgown.
Choice of containers/material handling
Spray drying facilities have a choice of handling methods and containers to use throughout the process for powder feed and receipt of starting and final materials. The most common choices are flexible/disposable Intermediate Bulk Containers (FIBC) and/or Rigid Intermediate Bulk Containers (IBC).
- FIBCs incur lower capital expenses and do not require cleaning apparatus or storage space between uses, but do have some volume limitations.
- Rigid IBCs are the most common and user-friendly option. Although they boast larger volumes capability, IBCs incur higher capital expenses, and they need to be washed between every use and stored accordingly.
Regardless of type of container, one of the key considerations in the design is the type of valving on the IBC inlet and/or outlet. Depending on the containment requirements and docking arrangement, this may take the form of standard butterfly valves, split butterflies or other flexible containment systems (i.e. continuous liners).
The future of spray drying
Over the decades, spray drying has proven itself to be a predictable and reliable process to create high quality pharmaceuticals. Exciting innovation in recent years may open the door to new areas of formulation and new novel drug delivery methods. In the meantime, if your team needs guidance on how to optimize your spray drying process—or wants help navigating the new possibilities—our team of seasoned professionals is ready to help.

A COVID-19 roadmap for oral solid dosage manufacturersOSD products may find themselves thrust into the spotlight as their effectiveness against the symptoms of COVID-19 becomes known.
Read More
OSD products may find themselves thrust into the spotlight as their effectiveness against the symptoms of COVID-19 becomes known.
Read More
