Comparing classic plasma fractionation separation methodsComparing classic plasma fractionation separation methods—and what comes next.
Read More
Comparing classic plasma fractionation separation methods—and what comes next.
Read More
The plasma fractionation process comes with many challenges. One, in particular, stands out from a facility engineering point-of-view: keeping the entire process cool from start to finish. This is essential to preserve the plasma and the separation agent, ethanol.
Maintaining low temperatures throughout the entire process requires:
Here’s what the process looks like from beginning to end.
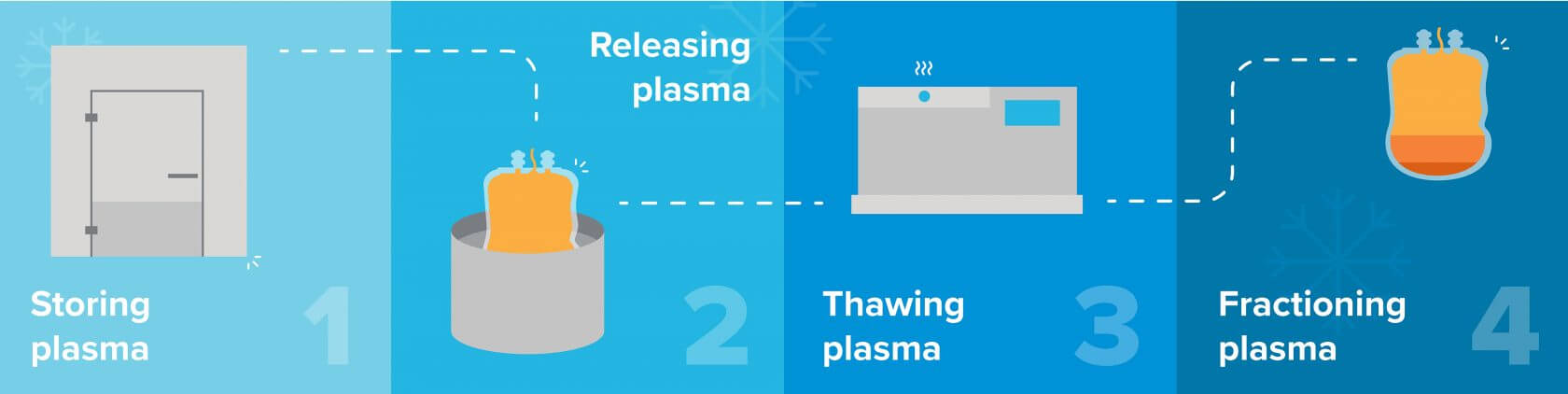

Plasma is kept in cold rooms (e.g. a walk-in freezer) for up to three years. It must be stored between -25℃ to -30℃ (-13℉ to -22℉). This requires an air-cooled condenser, such as a unit on the roof that absorbs heat from the refrigerated space and rejects it outside. From a safety point-of-view, cold rooms must have emergency latch releases so operators cannot get locked inside.
When the plasma comes out of storage to undergo the fractionation process, it must be released from its storage bag. It gets dunked in liquid nitrogen to make the bag brittle for easy removal, efficiently reducing the amount of wasted product. As such, fractionation facilities must keep a supply of liquid nitrogen on hand. It is typically stored in a tank outside the facility and piped into the building.
Next, the plasma is thawed in a large vessel. This process requires precision control, so you can’t rely on ambient temperature. Instead, you can dial in the exact temperature you need by wrapping the vessel in a jacket that recirculates heat transfer fluid that the chiller system supplies. This works like an insulated thermos that keeps a beverage chilled. The jacket contains a thin layer of liquid that draws heat from the plasma. The chiller controls the temperature of the liquid and a pump controls the flow, constantly recirculating the liquid to keep it cool, thereby controlling the overall temperature of the plasma inside the vessel. Once thawed, the plasma must be kept at 4℃ (39.2℉) and processed within 24 hours.
The thawed plasma is ready for fractionation. Ethanol is a key separation agent in this process, as it pulls apart the plasma’s proteins. To use ethanol in the precipitation steps, it needs to be stored at a high concentration. The challenge is that ethanol has a flashpoint of 13℃ (55℉), which is well below room temperature of about 23℃ (73℉). If it warms above its flashpoint, it can auto-ignite (i.e. no spark needed), so you must store ethanol below its flash point to reduce the chance of combustion. Keep your ethanol cool both during storage and when adding it to the plasma.
Additionally, throughout processing, you will need to spray down centrifuge bowls with cold ethanol to keep them cold. Also, cool down any buffers added into the plasma below the freezing point of water, which is possible due to the buffer composition.
Due to its low temperature requirements, the plasma fractionation process is difficult and places high demands on all the equipment. Furthermore, reliable equipment is absolutely critical to ensure safety of the operators and effectiveness of the product. If the plasma temperature falls out of the acceptable range during processing, then the entire brew-and-batch must be thrown out—a single incident might cost millions!
The system uses two key heat transfer thermofluids: a refrigerant contained in the chiller and a mixture of glycol and water that is distributed throughout the building (sometimes called a brine or simply referred to as glycol) to transfer the heat at various points in the process. In some situations, cooling the chiller itself might require a third thermofluid.
The chiller circulates refrigerant through four critical components: the evaporator, compressor, condenser, and metering device. The chiller system recirculates cold liquid for the purpose of heat transfer. The chiller controls the temperature throughout processing by transferring heat out of the very cold glycol solution and rejecting it either into water or into the air. If the heat is rejected into water, an outdoor cooling tower will, in turn, cool the water using outdoor air.
The cooler system we’re talking about needs to run very low temperatures: ranging from -10 ℃ (10℉) to -30 ℃ (-22℉). A chiller capable of discharge temperatures this low needs to be much more powerful than an equivalent tonnage system designed for building HVAC.
For example, one chiller might be able to supply a -14℃ (8.6℉) glycol mixture at 100 gallons per minute, whereas another can supply around 200 gallons per minute at 6.7℃ (44℉). The example is simplified, but the point remains that there are tradeoffs, like temperature versus volume. Equipment plays a significant role in low temperature heat transfer. In some cases, if the supply temperature is low enough, your system might require specialty equipment, such as a custom chiller. Keep in mind that a chiller also contains a lubrication system and many other subsystems which must be coordinated to allow it to create such a low supply temp for the glycol.
One more consideration for chillers: due to the nature of this application, the coefficient of performance (COP) and therefore the energy efficiency ratio (EER) are much lower than most chilled water systems. This means the horsepower per ton requirements are much higher than typical HVAC or even many other process applications. Compression ratio is significantly higher in this application than in most chiller applications. This increases the lift the compressor must generate, as well as increasing the input and waste energy. Basically, the plasma fractionation process requires bigger and more heavy-duty equipment to do the work because it is all done at significantly lower temperatures.
Because this chiller system needs to be colder than usual (below freezing), you might need a specialty refrigerant because plasma fractionation requires such low temperatures. For example, R-134a, a common refrigerant, is usually not suitable in a chiller supplying fluid temps below -13℃ (8℉).
Refrigerant selection influences the chiller design because it is typically difficult and costly—if not impossible—to convert a chiller to a different refrigerant. Therefore, you need to commit to your refrigerant early in the design process. Consult with the chiller manufacturer before you arbitrarily select a refrigerant, and make sure you consider the equipment already in use, and the serviceability, safety, and projected future availability of additional equipment. Environmental, toxicity, and combustibility (flammability) concerns and legislation may also constrain your refrigerant selection.
One final word of advice: refrigerant leaks can be damaging and costly. Make sure you actively monitor for leaks and make repairs immediately when they happen.
While the chiller controls the temperature, the pump powers the flow. The pump recirculates the liquid, or thermofluid, from the chiller through the whole building using a series of heat exchangers and vessel jackets.
Properly aligning and balancing a pump is critical to achieving reliability and longevity. A pump of this capacity is typically driven by a separate external electric motor, requiring a shaft seal to allow the input power to enter the pump without the glycol leaking out.
The piping forms the delivery system of the cooling system. Piping and manual and automatic valves within the pumps control the rate and direction of the glycol flow.
The combination of the low temperature fluid in the pipe and the dew point of the air outside of the pipe can cause condensation to form on the surface of the pipe, just like a cold glass of water on a hot day. Condensation can lead to dripping, which may create mold, slippery surfaces and ice blocks. To prevent this, insulate all exposed pipes. Select the right insulation with two things in mind: minimizing heat gain into the piping (which is a parasitic heat load) and ensuring the surface temperature of the insulation remains below the dew point of the surrounding air. A vapor barrier and sealed insulation sheathing prevents condensation from forming on the pipe, and the insulation prevents the surface of the insulation sheathing from being cold enough to form condensation. The insulation needs an air-tight seal.
A normal chilled water system uses only water, but in this case, you’ll need to use a heat transfer solution instead of chilled water to achieve heat transfer fluid temperatures low enough for plasma fractionation. The solutions generally include ethylene glycol or propylene glycol (mixed with water) which can bring the freezing point of the mixture to below -10℃ (14℉).
A few thousand gallons of the glycol water mixture run through the entire cooling system. The liquid is recirculated through a closed system and only replaced if it is spilled or in the event of facility construction. These fluids can be oily or toxic, so be aware of spills and make a clear plan to prevent fluids from entering the city sewer.
Additionally, there are complications that come with using glycol: biological growth control, increased corrosion, and lower heat transfer rates. Using glycol requires larger heat exchangers and higher flow rates, therefore requiring bigger pipes, bigger pumps, and higher pumping energy requirements compared to water. Chemical additives packages (i.e. inhibitors and buffers) must be added to the glycol to inhibit biological growth and corrosion. Also, perform scheduled, periodic monitoring and chemical testing on the glycol to ensure it is not degrading.
Bring the system online slowly to prevent rapid temperature changes. It may take hours to bring the system fully online and achieve low enough temperatures. Rushing the process may cause the system to fail. Finally, be prepared to measure and adjust flow rates throughout the entire system during the commissioning process and the testing and balancing (TAB) phase.
Every part of this cooling system uses instrumentation to monitor temperatures, pressures, and flow rates. This is all controlled by the building automation system (BAS), which tells the chiller or pump when to turn on and the valves when to open (or how much to open if they have modulation).
The BAS is also responsible for detecting a failure and energizing redundant equipment, equalizing runtime hours, starts and stops, lead/lag control, etc. In a facility that has chillers and other components of unequal sizes and unequal capacities, the automation system may also select which piece of equipment most closely matches the load.
In addition to needing more powerful and sometimes specialized equipment, the reliability of the plasma fractionation process equipment is critical to the success of the process. This is where redundancy equipment comes in. In the same spirit as the Boy Scout motto of “Be prepared,” standby equipment will ensure that an equipment failure is merely an inconvenience rather than a disaster.
While piping typically has a lifespan of many decades, equipment with rotating and moving parts (e.g. chillers and pumps) wears out more quickly and may unexpectedly fail. The only way to keep it running is with redundant equipment that can be turned on at a moment’s notice. Standard practice dictates that critical components are fully redundant with a completely capable standby unit piped in parallel ready to carry the load the second it’s needed.. For example, keeping a chiller on hot standby may involve the use of heaters for the lubricating oils and other devices to ensure it’s ready to start immediately if the main chiller fails. If a pump fails, the BAS software will instantly detect it via a failure alarm or pressure problem and it will automatically turn on the redundant one.
This approach requires ordering at least two of any major pieces of equipment (N+1, N=minimum required) and then connecting the equipment in parallel or series as necessary to achieve the redundancy required.
Cold temperatures present a number of challenges for different materials. In terms of material durability as temperatures fall, many materials (eg. plastic) become more brittle and less ductile. Also, cold surfaces—not just the piping—need to be insulated to prevent condensation, which leads to corrosion and dripping. Stainless steel holds up against corrosion and performs well in the cold. Also, select gasket material with the cold in mind; nitrile and EPDM are good choices.
Plasma fractionation is a unique and highly demanding process you may need a custom chiller to meet your facility’s needs. Low temperature chillers are harder to come by than chillers that generate the warmer supply temperatures used in common applications, such as for facility air conditioning. Be aware that custom pieces often come with big price tags and long lead times.
There are usually two to four chiller systems to serve a low temperature production line and within a plasma fractionation facility, there may be multiple systems to accommodate different temperatures. For example, a single building could have separate chiller systems operating at 6℃ (43℉), -10℃ (14℉), and -20℃ (-4℉) to accommodate all the cooling requirements for the process. Another way to accommodate different temperature requirements for various parts of the process is to have a sub-loop fed off a main system that then returns back into that loop. This approach uses the same chiller but a different pump and distribution. Analyze these options during the design stage of your facility.
Excellent planning is crucial to maintaining the kind of low temperatures required throughout the entire plasma fractionation process. Altering thermal capacity, flow rates, supply temps, or component pressure loss may require significant design updates or even a complete rework of the design. Therefore, you must decide on these factors early in the design process. Also, if flexibility or future growth are your goals, communicate this to the design team early in the process.
Another consideration is the ability of the equipment, especially including the chiller, to operate at less than full heat load conditions. Consider the diversity of load/peak load, as well as part load operating efficiencies.
Ultimately, if you want to keep your plasma fractionation process and facility consistently and adequately cool, remember the general guideline that capital cost only represents 20% of equipment total cost over its lifespan. This type of equipment is not the place to cut corners. It will serve as the underpinning of the entire operation and its reliability is key to profitability.
Comparing classic plasma fractionation separation methods—and what comes next.
Read More