The COVID-19 pandemic has put the spotlight on the FDA drug approval process. Why will it take so long for a vaccine to get approved?
Recently, the World Health Organization said there are around seven or eight “top” candidates for a vaccine to combat the novel coronavirus and work on them is being accelerated. This includes several major firms, such as Johnson & Johnson, Pfizer, and Moderna. AstraZeneca announced that it will be ready to mass-produce a COVID-19 vaccine by September 2020. However, the pharmaceutical company acknowledges that it has not yet confirmed that the vaccine is effective. That will require a series of clinical trials—a lengthy, but necessary process.
The pressures on the pharmaceutical industry and global regulatory bodies are ever-increasing. The entire world wants and needs to see quick identification and development of vaccine candidates and new drug therapies, along with the regulatory approvals, necessary to end this pandemic. And the world wants to see it NOW.
Unfortunately, the drug development and approval process was not designed for speed. It was designed for safety. To put the task at hand into perspective and offer those not knowledgeable of the approval process, this is an overview of the current Food and Drug Administration (FDA) Drug Approval Process and what it takes to bring a drug to market. It will make clear the challenges we are faced with to accelerate the process.
Drug approval takes time and money
According to the Tufts Center for the Study of Drug Development, the full research, development and approval process for a new drug product can take from 12 to 15 years. The investment made by the drug manufacturer to get a new drug from lab bench to pharmacy exceeds $2.6 billion dollars. Only one in 1000 of the compounds that enter laboratory testing ever make it to human testing and only a small percentage of those make it to the market.
The drug approval process
Scientists around the world are fully dedicated to finding cures and treatments for the wide range of diseases that threaten humanity. In our history, there have been a remarkable number of breakthroughs, and a large number of diseases are now treatable or have been beaten. As I write this brief, I am optimistic that COVID-19 will be on that list soon. But what does it take and how does approval occur? Let me answer a few of the basic questions.
What does the FDA define as a Drug?
A drug is any product that is intended for use in the diagnosis, cure mitigation, treatment, or prevention of a disease and that is intended to affect the structure or any function of the body. When a drug candidate is discovered, it must go through the FDA New Drug Approval (NDA) submission process.
Who at the FDA reviews the new drug submissions?
A new drug submission is reviewed by an experienced team of scientists, chemists, pharmacologists, statisticians, and physicians. Each group reviews the drug sponsor’s data and labeling claims, all with an eye toward safety and efficacy.
How does the drug approval process progress and what are the key stages?
There are three primary phases of the approval process: pre-clinical trials, clinical trials, and new drug application review.
Pre-Clinical Trials:
This is where a drug sponsor (usually a pharmaceutical company) develops a new compound and seeks to have it approved by the FDA for sale in the United States. The sponsor must test the drug on various animal species for toxicity to generate data and information on the safety and efficacy of the compound being investigated.
When the data is encouraging, the sponsor submits the formal request to the FDA. This is an Investigational Drug Application (IND). It must include the pretesting details, the drug’s composition, and a plan for testing the drug on humans.
The FDA ensures that the data and proposed studies address safety and do not put human subjects at an unreasonable risk of harm.
Pre-clinical research and trials to get a working compound can take many many years.
Clinical Trials:
With an approved IND the drug sponsor can proceed to clinical trials in human beings. There are three distinct phases of this process that typically run sequentially. These include:
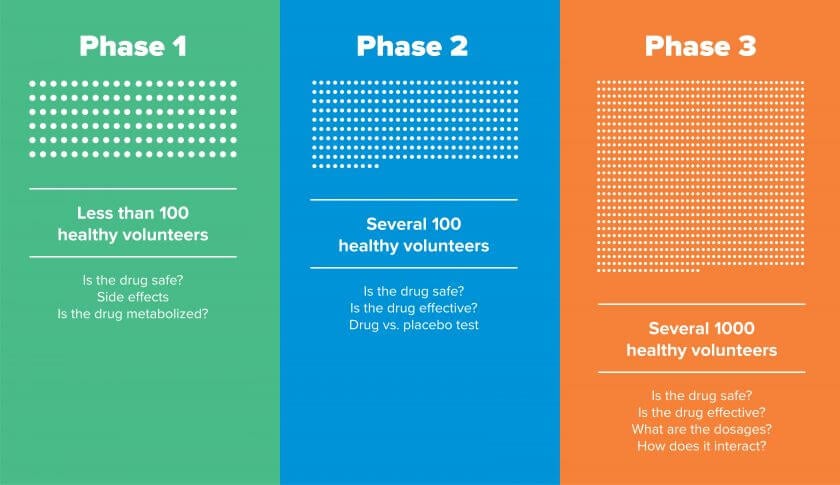
Phase I:
A relatively small sample of healthy volunteers (typically <100) are given the drug. The goal here is safety. Phase I reviews the most frequent side effects and determines how the drug is metabolized and excreted. If successful, you can progress to phase II. If not, the trial ends.
Phase II:
A larger sample of patients who have a certain disease or condition (typically in the 100s) is given the drug. The goal of phase II is to confirm that the drug is effective. This phase is a controlled study that includes a comparison of results from those given the actual drug, a non-active placebo, or a different drug. Safety and side effects are further evaluated. If successful, you can progress to phase III after additional discussion and agreement on how the larger scale trials will be completed. If not, the trial ends.
Phase III:
A large sample of patients (typically in the 1000s) is given the drug. These trials gather more information on the safety and effectiveness of the drug across different populations and dosages, and in combination with other drugs. If successful, you can progress to the New Drug Application (NDA) Review and selling approval. If not, the trial ends.
Getting through all three phases of the clinical trials has historically taken an average of three years or more. Under the current crisis, the FDA is allowing for acceleration of this timeline and some trials to overlap as opposed to running sequentially.
New Drug Application Review:
With successful clinical trials the sponsor can progress to an NDA review process that ultimately leads to being able to market and sell the product for use. This includes multiple steps that involve:
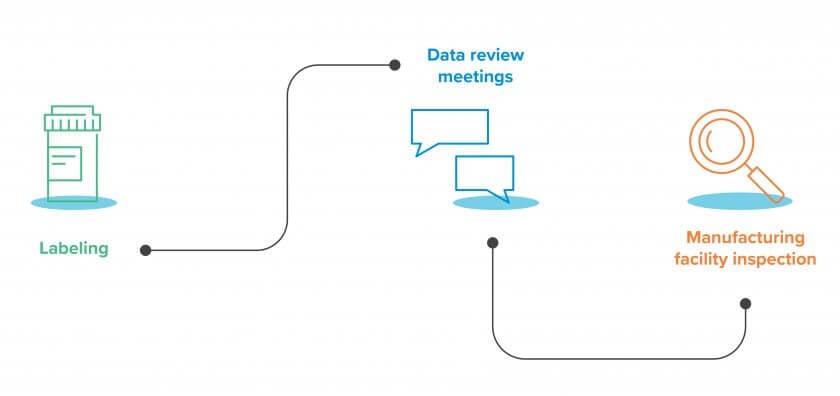
Drug labeling:
The agency reviews the labeling to ensure that it adequately and accurately communicates necessary information about the drug to health care professionals and consumers. This review process also verifies that false claims are not being made in the labeling of all end products.
NDA review/application review and sponsor meetings:
This includes a multi-step process that incorporates detailed reviews of the sponsor’s research, clinical safety data, animal and human analysis data, as well as meetings with the sponsor.
Manufacturing facility inspection:
Prior to the final approval, the FDA will inspect the manufacturing facility to ensure it’s compliant with current Good Manufacturing Practices (cGMP) and it’s safe.
After a drug successfully completes all these steps, the FDA grants drug approval and the drug product can be supplied to the market.
Additionally, there is a formal fourth phase: a Post Marketing and Post Approval Risk Assessment. This monitors the drug’s safety after it is on the market and detects any serious, unexpected adverse events that were not possible to predict during the clinical trials. The sponsor submits periodic updates to the FDA.
To get to an approved drug product, the timetable is typically not quick and the sponsor incurs a high cost. Further, a high percentage of products never make it through the process to approval. Fortunately, the FDA does offer an Accelerated Approval Program and a Fast Track Program to speed up the process which is occurring today under the COVID-19 pandemic.
Fast Track:
This process facilitates rapid development and expedites the review of drugs that treat serious conditions and/or fill an unmet medical need.
Accelerated Approval:
For drugs that address serious conditions and fill an unmet medical need, using a surrogate endpoint enables the FDA to approve these drugs faster.
Priority Review:
This designation means the FDA will endeavor to take action on an application within six months.
This will have to apply to the COVID-19 pandemic. However, there is no magic bullet, and as urgent the need may be, we still have to rely on the clinical trials. A safe and effective vaccine or treatment is more important than a fast vaccine that doesn’t work.
Working together with governments, global health organizations and regulators, COVID-19 will be addressed and we as a pharmaceutical industry will be smarter and stronger for it. Stay safe…stay healthy!
COVID-19 drug development strategiesCOVID-19 drug development strategies for the near-term, medium-term and long-term.
Read More
COVID-19 drug development strategies for the near-term, medium-term and long-term.
Read More
Keys to accelerating your COVID-19 drug campaignAt CRB, we have the experience, the tools, and the flexibility to help your facility respond to pandemic response production to reliable, commercial-scale capacity as fast as possible.
Read More
At CRB, we have the experience, the tools, and the flexibility to help your facility respond to pandemic response production to reliable, commercial-scale capacity as fast as possible.
Read More
